As the most commonly used color phosphor in w-LEDs, Ce: Y3Al5O12 (Ce: YAG) makes an almost perfect match with blue chips to efficiently convert the blue light into yellow light and obtain white light. But unfortunately, the deficiency of red component in the mixed white light makes the light quality too poor to meet the standard of modern lighting. The redshift of Ce3+ emission in Ce: YAG has always been the pursuit of industry and researchers.
Recently, a research team from Shanghai Institute of Optics and Fine Mechanics, Chinese Academy of Science, fabricated a new composite phosphor ceramic by introducing ZrO2 as the second phase into Ce: YAG. The result was published in Journal of the European Ceramic Society.
In their experiment, commercial Y2O3, α-Al2O3, CeO2, ZrO2 powders were used as raw materials. The powders were weighted according to the formula and were fully mixed. After ball milling in ethanol for 24 h, the slurry was dried at 75 oC and granulated with a 200-mesh sieve. After pressed into disks under uniaxial pressure of 10 MPa, the disks were processed under cold isostatic pressing at 200 MPa. The pressed disks were then heated in a muffle oven at 700 oC for 3 h to remove the organic ingredients. Finally, the phosphor ceramic samples were obtained after sintered at the temperature of 1650 oC and 1700 oC for 12 h, respectively.
A novel redshift mechanism of Ce3+ emission was discovered in this system, by which the emission wavelength of Ce3+ could be largely redshifted from 540 nm to 570~610 nm. This mechanism of redshift is argued to be related to the non-radiative transition of the oxygen vacancy energy level brought by the occupation of Zr3+ in the dodecahedral site.
The microstructure, luminescence properties, and the performance of ZrO2-Ce: YAG based w-LED devices were also investigated thoroughly.
This kind of redshift provides a new approach to the supplement of the red component in Ce3+ emission spectra beyond the regular ways of larger rare-earth ions doping or M2+-N4+ double substitution.
This work was supported by Innovation Program of Science and Technology Commission of Shanghai Municipality and the International Partnership Program of Chinese Academy of Sciences.
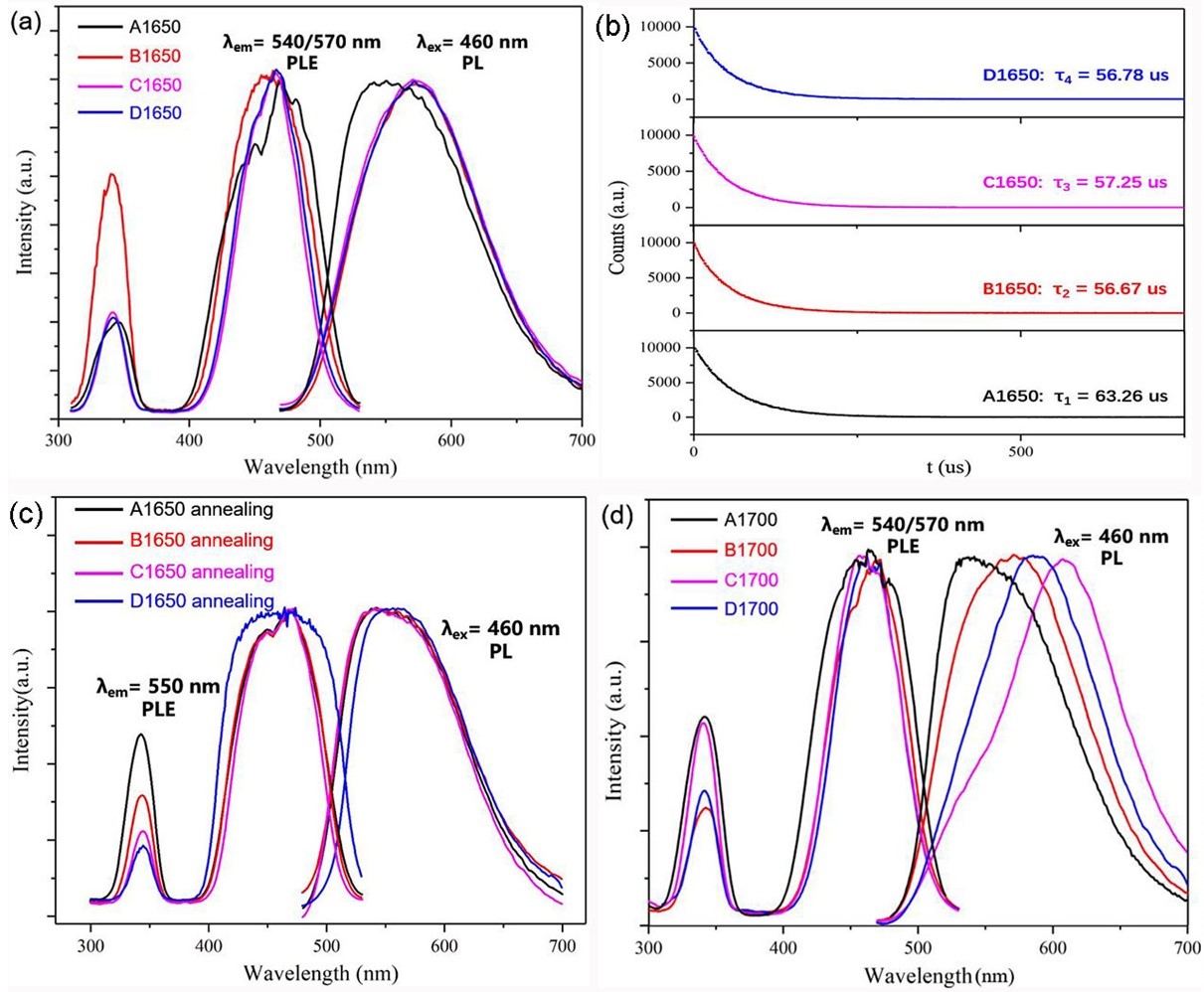
(a) The PL and PLE spectra of A1650-D1650; (b) The luminescence decay profile of A1650-D1650; (c) The PL and PLE spectra of A1650-D1650 after annealing in the air; (d) The PL and PLE spectra of A1700-D1700. (Image by SIOM)
Article website:
https://doi.org/10.1016/j.jeurceramsoc.2020.06.026
Contact:
Mr. CAO Yong
General Administrative Office
Shanghai Institute of Optics and Fine Mechanics, CAS
Email: caoyong@siom.ac.cn