The Ce:(Y1-x, Mgx)3(Al1-y, Siy)5O12 transparent ceramics have been broadly studied for the application in white light emitting diodes for its excellent mechanical, optical properties, and the large red component in its spectra. A stronger crystal field splitting and red shift emissions of Ce3+ can be accomplished through the introduction of Mg2+-Si4+. In fact, due to similar ionic radius of Mg2+ with Y3+ and Al3+, both sites of Y3+ and Al3+ can be substituted by Mg2+. However, the analysis of the structure and luminescence for Mg2+ doped in different sites are still unclear.
Recently, researchers from Shanghai Institute of Optics and Fine Mechanics of Chinese Academy of Science (SIOM, CAS) have reported a comprehensive structural study of the distinct red-shift properties of Ce:YAG transparent ceramics induced by Mg2+ doping in different coordinative sites. The results were published in Journal of Alloys and Compounds.
In the studies, the Mg2+-Si4+ co-doped in Ce:YAG ceramic phosphors were fabricated through the vacuum sintering technology. Because both MgO and SiO2 are good co-solvent, the sintering temperature decreased from 1700℃ to 1600℃, with the increase of Mg2+-Si4+ pairs. Therefore, it is an advantage to save costs. Then, the structure for Mg2+ doped in Y3+and Al3+ was discussed according to the Rietveld refinements and Raman spectra. The different changing rules of lattice constants and shifting rules of Raman peaks for Mg2+ doped in Y3+ and Al3+ were ascribed to the bond length and bond strength of [Mg-O].
Furthermore, the luminescence properties were investigated by the photoluminescence and photoluminescence excitation spectra. Different emission peaks shifting for Mg2+ doped in Y3+ and Al3+ was explained by means of the configuration coordination model. It was illustrated that the main reason for the emission red-shift was Stokes shift.
This paper firstly discussed the effects of Mg2+ doped in Y3+ and Al3+ site on the structure and luminescence, which is of great significance for the future research.
The work was supported by the International Partnership Program of Chinese Academy of Sciences and National Key R&D Program of China.
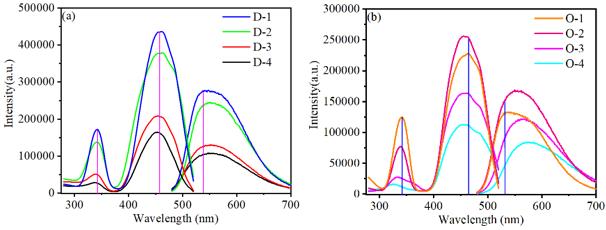
Fig.1 PLE and PL spectra of MYASG samples and YMASG samples. (Image by SIOM) 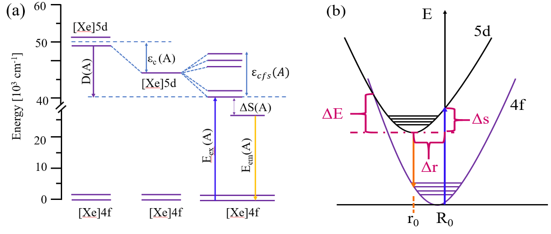
Fig.2 The energy level schemes of Ce3+ as free ion and in compounds A (a). The configurational coordinate diagram of YAG (b). (Image by SIOM)
Article website:
https://doi.org/10.1016/j.jallcom.2019.152236
Contact:
Mr. CAO Yong
General Administrative Office
Shanghai Institute of Optics and Fine Mechanics, CAS
Email: caoyong@siom.ac.cn